ISO 10993-1 Risk-Based Biological Evaluation
The biological evaluation process under ISO 10993-1 aligns seamlessly with ISO 14971 risk management principles, addressing biological safety through risk analysis, evaluation, control, residual risk assessment, and post-market activities. ISO 10993-1 is no longer a simple checklist. Instead, it provides a systematic framework to identify potential chemical, physical, and biological hazards associated with a medical device, perform gap analyses against predicate devices to identify biological equivalence, determine the need for biocompatibility testing, and implement risk control and monitoring throughout the product life cycle.
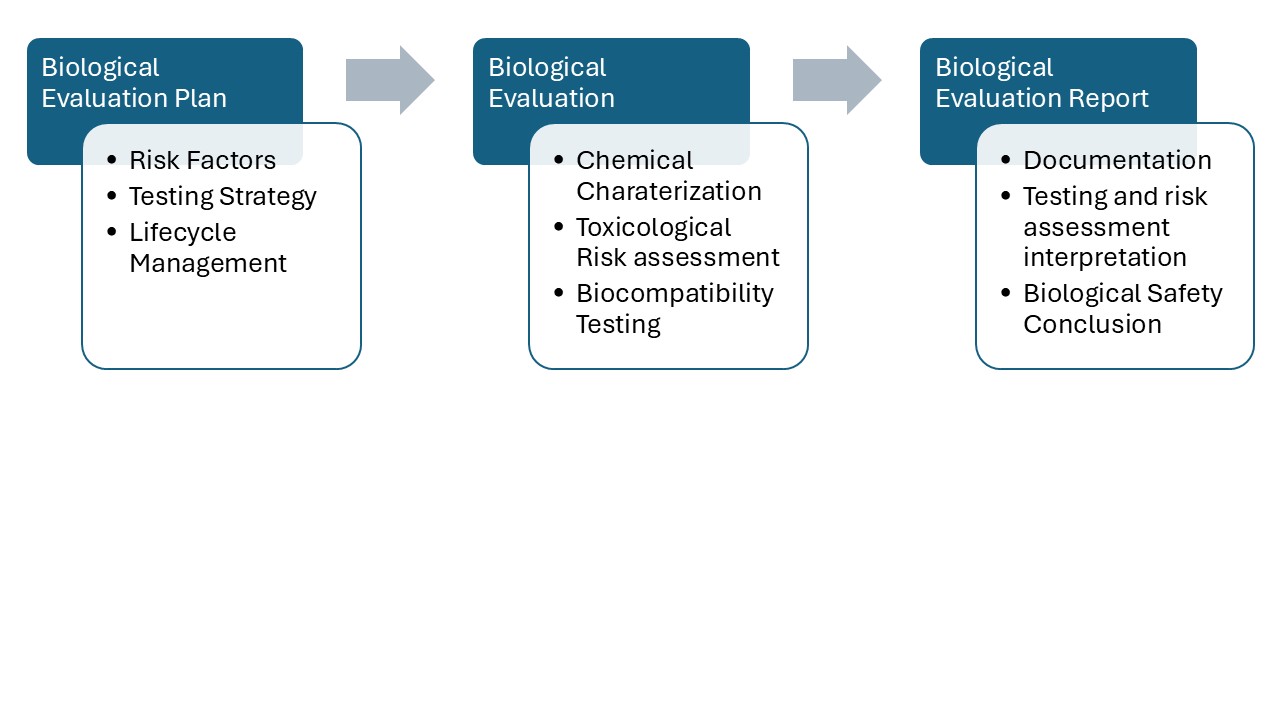
Developing a Biological Evaluation Plan (BEP)
Regulatory compliance and biological safety begin with a well-structured BEP. Establishing a BEP early in device development helps minimize biological risks and avoid unnecessary testing. Using available scientific data and device-specific knowledge, Medgaea offers strategic expertise in developing BEPs with a strong focus on regulatory compliance for clients. We construct BEPs that consider
- Material composition and physicochemical properties
- Intended use and reasonably foreseeable misuse
- Contact type and duration
- Identification of potential hazards and harms
- Gap analysis of predicate devices
- Determination of required biocompatibility testing
Preparing a Biological Evaluation Report (BER)
Following the BEP, a BER documents the findings and conclusions that demonstrate the device’s biological safety. The report also serves as a comprehensive record within the lifecycle risk management and can be updated as the product changes. Medgaea assists in preparing BERs in accordance with ISO 10993-1, aligning the scientific rationale with the technical documentation. Key components include
- Device description summaries consistent with product specification, materials, and manufacturing processes
- Regulatory-focused interpretation of biocompatibility test results
- Summaries of chemical characterization and toxicological risk assessment
- Risk assessment justification for testing exemptions or the use of reference data
- Overall evaluation on biological safety
At Medgaea, we deliver biological evaluations that are science-driven, regulatory-focused. Our expertise helps clients demonstrate device safety justifications and prepare supporting documentation. This ensures compliance with stringent regulatory requirements worldwide and accelerates product approvals and market entry in the medical device industry worldwide.