ISO 10993-17 Toxicological Risk Assessment
Toxicological risk assessment (TRA) under ISO 10993-17 provides a structured framework to evaluate the safety of chemical constituents in medical devices. This evaluation goes beyond a simple yes-or-no determination. It combines quantitative and qualitative assessments of potential health risks based on toxicological evidence and estimated exposure levels. Supporting TRA, chemical characterization (ISO 10993-18) plays a pivotal role in identifying potential chemical risk factors at the early stages of biological evaluation.
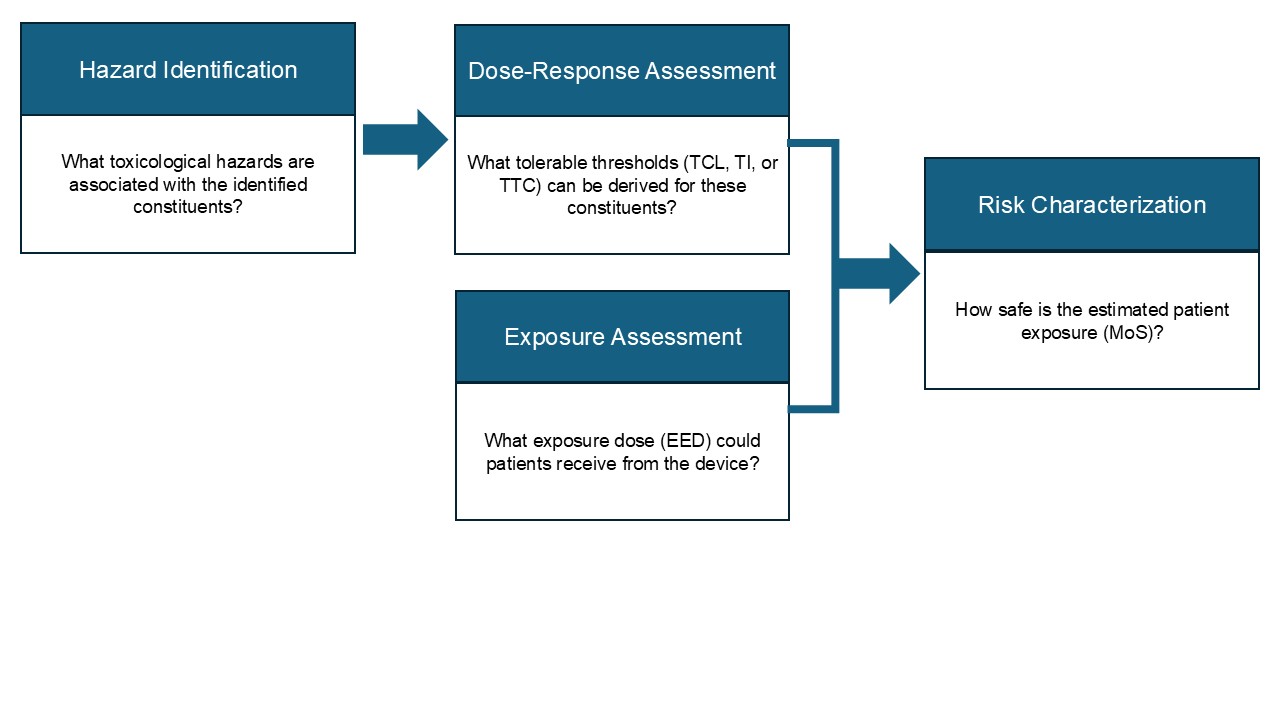
The risk-based principles emphasized in ISO 10993-1 further reinforce the importance of TRA for managing chemical risks and protecting patient safety. At Medgaea, we follow the systematic 4-step approach to risk assessment under ISO 10993-17:
- Hazard identification: Review international toxicological databases for identified constituents to determine potential harms and whether these harms could occur under human exposure.
- Dose-response assessment: Establish toxicological thresholds (e.g., tolerable intake, tolerable contact level) to evaluate the relationship between exposure and the likelihood or severity of harm.
- Exposure assessment: Estimate the magnitude of patient exposure under worst-case medical device use scenarios.
- Risk characterization: Integrate the findings from the previous steps and derive a Margin of Safety (MoS) to support risk-based conclusions regarding device safety.
Following the 4-step risk assessment process, Medgaea provides scientific-based rationale to define acceptable toxicological risks. Our team applies professional judgement to evaluate chemical-specific toxicological profiles and conducts risk-based evaluations to establish safe exposure limits for device constituents. This process helps clients demonstrate safety justifications and meet the requirements of stringent regulatory reviews.