ISO 10993-18 Chemical Characterization
Obtaining chemical information is one of the critical steps at the beginning of biological evaluation process. Substances associated with the device, including materials of construction, processing aids, and residual contaminants from manufacturing process, may represent potential hazards. Chemical characterization addresses three key questions through a scientific evaluation:
- Constituent identification: What chemicals are present in the device?
- Quantitative analysis: How much of each chemical could patients be exposed to?
- Exposure assessment: Could patient exposure pose health risks during intended use?
These questions also form the basis for toxicological risk assessment (ISO 10993-17).
Systematic Evaluation Process and Strategies:
Chemical characterization identifies materials of construction and profiles device constituents to determine what may be released or migrate during clinical use. By combining advanced analytical techniques with toxicological databases, chemical characterization can also identify the presence of cohorts of concern (CoC) (refer to ISO 21726), which are chemicals with high toxicological potency, such as human carcinogens.
To improve toxicological risk assessments (TRAs), risk-based chemical characterization strategies include
- Threshold setting: Establishing dose-based thresholds (DBTs), such as the toxicological threshold of concern (TTC).
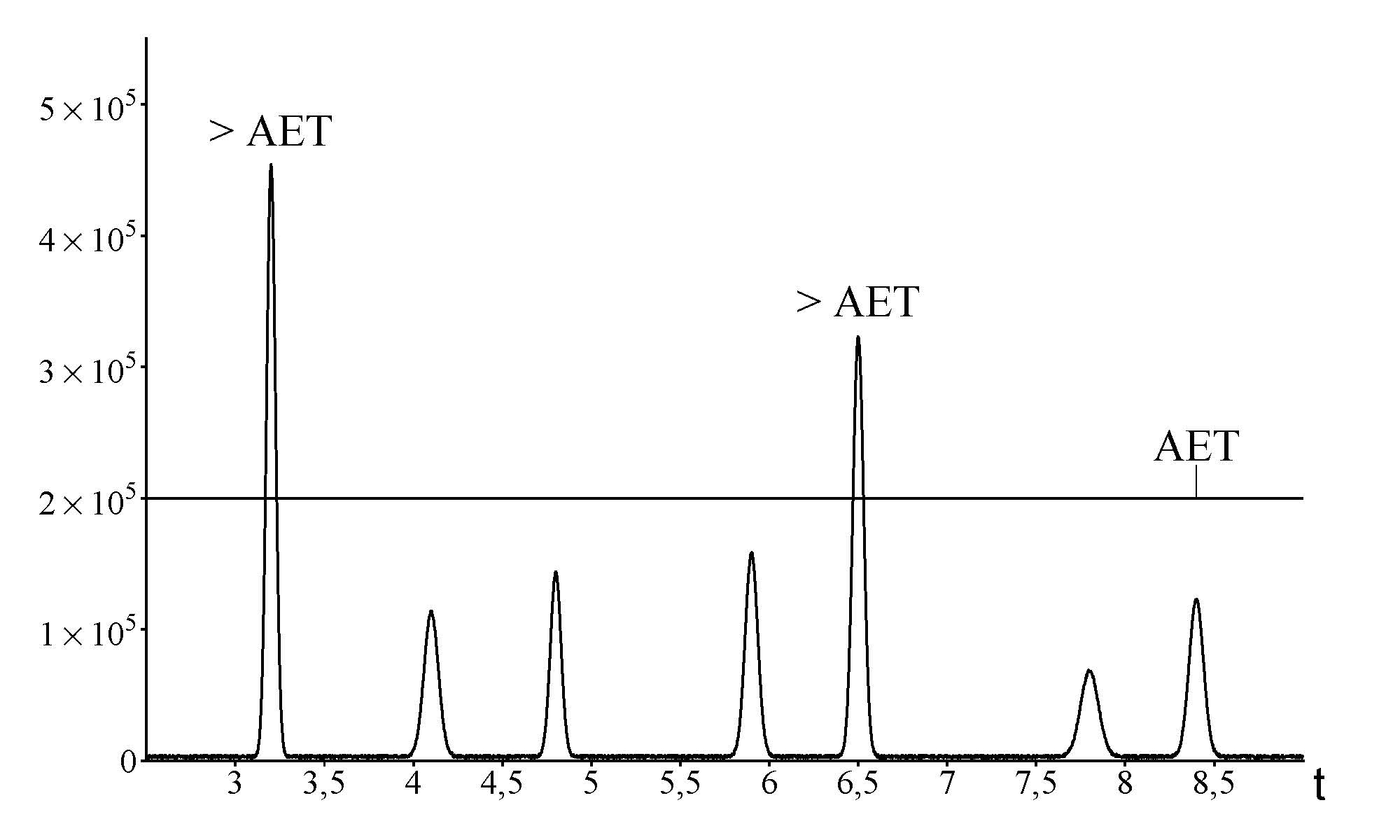
- Analytical Evaluation Threshold (AET) calculation: Using AET as a screening baseline to determine which detected compounds require further identification and quantification for TRAs.
- Systematic detection: Characterizing extractables and leachables (E&L) to assess potential chemical exposure and ensure biological safety.
This risk-based approach prioritizes the most clinically relevant constituents that may pose potential hazards. It supports efficient TRAs and helps achieve a high level of biocompatibility for medical devices
AET application on chromatographic analysis. E&L above AET should be identified and reported for toxicological risk assessment.